Porsche "Variable turbine geometry"
"The 997 911 Turbo uses a BorgWarner VTG turbocharger which uses special materials derived from aerospace technology, hence solving the temperature problem."
humm sounds like Dr. Porsche broke out the.. http://en.wikipedia.org/wiki/Iridium

Industrial and medical
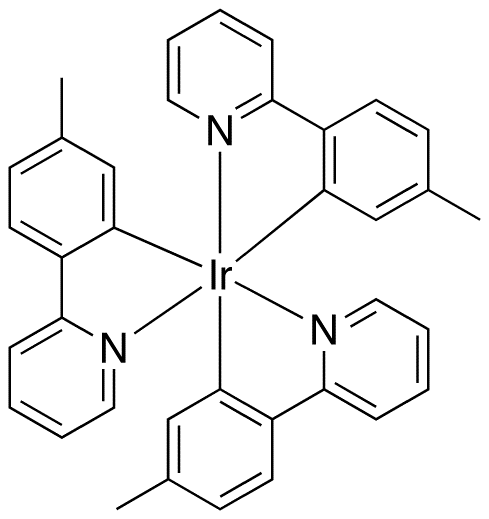
Molecular structure of Ir(mppy)3The high melting point, hardness and corrosion resistance of iridium and its alloys determine most of its applications. Iridium and especially iridium–platinum alloys or osmium–iridium alloys have a low wear and are used, for example, for multi-pored spinnerets, through which a plastic polymer melt is extruded to form fibers, such as rayon.[53] Osmium–iridium is used for compass bearings and for balances.[10]
Corrosion and heat resistance makes iridium an important alloying agent. Certain long-life aircraft engine parts are made of an iridium alloy and an iridium–titanium alloy is used for deep-water pipes because of its corrosion resistance.[10] Iridium is also used as a hardening agent in platinum alloys. The Vickers hardness of pure platinum is 56 HV while platinum with 50% of iridium can reach over 500 HV.[54][55]
Devices that must withstand extremely high temperatures are often made from iridium. For example, high-temperature crucibles made of iridium are used in the Czochralski process to produce oxide single-crystals (such as sapphires) for use in computer memory devices and in solid state lasers.[56][57] The crystals, such as gadolinium gallium garnet and yttrium gallium garnet, are grown by melting pre-sintered charges of mixed oxides under oxidizing conditions at temperatures up to 2100 �C.[4] Its resistance to arc erosion makes iridium alloys ideal for electrical contacts for spark plugs.[57][58]
Iridium compounds are used as catalysts in the Cativa process for carbonylation of methanol to produce acetic acid.[59] Iridium itself is used as a catalyst in a type of automobile engine introduced in 1996 called the direct-ignition engine.[10]
The radioisotope iridium-192 is one of the two most important sources of energy for use in industrial γ-radiography for non-destructive testing of metals.[60][61] Additionally, 192Ir is used as a source of gamma radiation for the treatment of cancer using brachytherapy, a form of radiotherapy where a sealed radioactive source is placed inside or next to the area requiring treatment. Specific treatments include high dose rate prostate brachytherapy, bilary duct brachytherapy, and intracavitary cervix brachytherapy.[10]
Refractory metals are a class of metals that are extraordinarily resistant to heat and wear. The expression is mostly used in the context of materials science, metallurgy and engineering. The definition of which elements belong to this group differs. The most common definition includes 5 elements, two of the fifth period niobium and molybdenum and three of the sixth period tantalum, tungsten and rhenium. They all share some properties, for example they all have a melting point above 2000 �C and are hard at room temperature. They are chemically inert and have a relatively high density. Their high melting points make powder metallurgy the method of choice for fabricating components from these metals. Some of their applications include tools to work metals at high temperatures, wire filaments, casting molds, and chemical reaction vessels in corrosive environments. Partly due to the high melting point, refractory metals are stable against creep deformation to very high temperatures.
humm sounds like Dr. Porsche broke out the.. http://en.wikipedia.org/wiki/Iridium


Industrial and medical
Molecular structure of Ir(mppy)3The high melting point, hardness and corrosion resistance of iridium and its alloys determine most of its applications. Iridium and especially iridium–platinum alloys or osmium–iridium alloys have a low wear and are used, for example, for multi-pored spinnerets, through which a plastic polymer melt is extruded to form fibers, such as rayon.[53] Osmium–iridium is used for compass bearings and for balances.[10]
Corrosion and heat resistance makes iridium an important alloying agent. Certain long-life aircraft engine parts are made of an iridium alloy and an iridium–titanium alloy is used for deep-water pipes because of its corrosion resistance.[10] Iridium is also used as a hardening agent in platinum alloys. The Vickers hardness of pure platinum is 56 HV while platinum with 50% of iridium can reach over 500 HV.[54][55]
Devices that must withstand extremely high temperatures are often made from iridium. For example, high-temperature crucibles made of iridium are used in the Czochralski process to produce oxide single-crystals (such as sapphires) for use in computer memory devices and in solid state lasers.[56][57] The crystals, such as gadolinium gallium garnet and yttrium gallium garnet, are grown by melting pre-sintered charges of mixed oxides under oxidizing conditions at temperatures up to 2100 �C.[4] Its resistance to arc erosion makes iridium alloys ideal for electrical contacts for spark plugs.[57][58]
Iridium compounds are used as catalysts in the Cativa process for carbonylation of methanol to produce acetic acid.[59] Iridium itself is used as a catalyst in a type of automobile engine introduced in 1996 called the direct-ignition engine.[10]
The radioisotope iridium-192 is one of the two most important sources of energy for use in industrial γ-radiography for non-destructive testing of metals.[60][61] Additionally, 192Ir is used as a source of gamma radiation for the treatment of cancer using brachytherapy, a form of radiotherapy where a sealed radioactive source is placed inside or next to the area requiring treatment. Specific treatments include high dose rate prostate brachytherapy, bilary duct brachytherapy, and intracavitary cervix brachytherapy.[10]
Refractory metals are a class of metals that are extraordinarily resistant to heat and wear. The expression is mostly used in the context of materials science, metallurgy and engineering. The definition of which elements belong to this group differs. The most common definition includes 5 elements, two of the fifth period niobium and molybdenum and three of the sixth period tantalum, tungsten and rhenium. They all share some properties, for example they all have a melting point above 2000 �C and are hard at room temperature. They are chemically inert and have a relatively high density. Their high melting points make powder metallurgy the method of choice for fabricating components from these metals. Some of their applications include tools to work metals at high temperatures, wire filaments, casting molds, and chemical reaction vessels in corrosive environments. Partly due to the high melting point, refractory metals are stable against creep deformation to very high temperatures.
Last edited by nyqueenz; Jun 27, 2010 at 08:57 AM.
well I'm also lookin into this myself as maybe an upgrade or maybe going custom in the future. But as far as I know I think Porsche came up with their own design wit the electro motors ver.. but like what i see in my research.. this technology has been around for years wit diesels..
What kind of maintenance would you need on these? You thinking they would have to be rebuilt after say 60k, based on driving style? With all that aerospace material being used, I figure they would last a long time, or so hope.
Thread
Thread Starter
Forum
Replies
Last Post
johnadlertech
Accessories
6
Sep 1, 2015 08:15 PM
OvaYahead
Engine, Drivetrain & Forced-Induction
11
Jun 3, 2003 07:28 AM